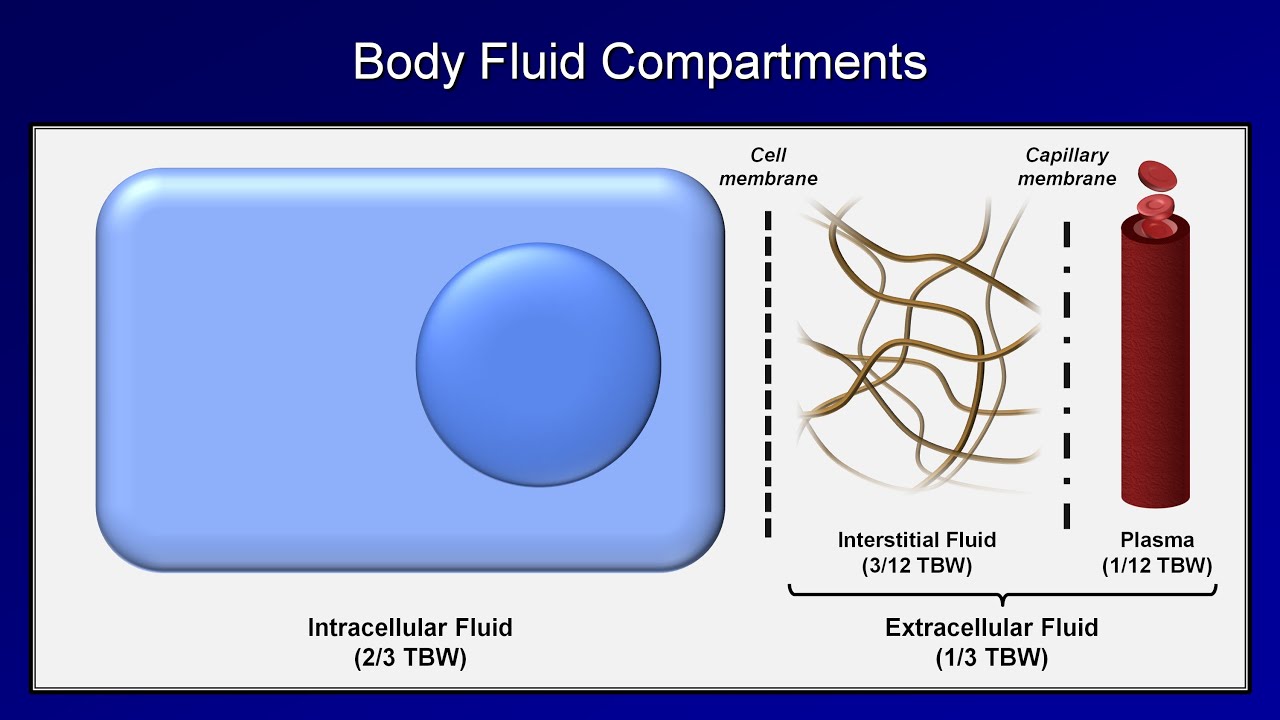
So for example, the K+ in the intracellular fluid is balanced out by negatively charged proteins and organic phosphates. The major intracellular cations are potassium (K+) and magnesium (Mg2+), whereas the major anions are proteins and organic phosphates like ATP.įluid compartments always maintain the same concentration of positive charges as negative ones in order to stay electrically neutral - that’s called the principle of macroscopic electroneutrality. Intracellular fluid is important for dissolving cations which are molecules with a positive charge, and anions which are molecules with a negative charge. Total body water can be subdivided into two major compartments, intracellular fluid which is fluid inside cells, and extracellular fluid which is fluid outside of cell like in the blood and in the interstitial tissue between cells.Īssuming that the total body water is about 60% of their body weight, roughly 2/3 of that, or 40% is intracellular fluid, and the other 1/3 or 20% is extracellular fluid. So a really muscular and lean person would have a relatively high proportion of their body weight made up of water.Īdditionally, females tend to have more fat than males and so on average tend to have lower proportion of their body weight made up of water. Since fat doesn’t store any water, a person’s water content is inversely proportional to a person’s fat content. The human body has 3 main mechanisms to control a change in the acid-base balance of body fluids: (A) Extracellular and intracellular buffering, (B) changing the respiration rate of the lungs and (C) adjustments to renal acid secretion.Water is the key to life, and it takes up a big proportion of our body weight, typically around 60 percent! The precise amount of water depends on a person’s body composition. 
Alkalosis can be caused by two mechanisms: (A) Respiratory Alkalosis and (B) Metabolic Alkalosis.Ĭompensation for a Disruption in the Acid-Base Balance Disturbance in the normal values of bicarbonate and pH lead to acidosis and alkalosis.Īcidosis and Alkalosis: Acidosis is a state of increased acidity in the body, characterized by a pH 7.45. The main function of this enzyme is to interconvert carbon dioxide and bicarbonate to maintain the acid-base balance in the blood and tissues.ĭavenport Diagram: The Davenport diagram is a graphical representation of the relationship between the plasma bicarbonate concentration and the pH of the blood. Henderson-Hasselbalch equation: The Henderson-Hasselbalch equation derives the pH as a measure of the acidity in biological systems.Īcid Formation in the Body: Carbonic Anhydrase is a metalloenzyme that rapidly and reversibly converts carbon dioxide and water into carbonic acid. Nephrons are connected via the collecting duct system fluid flows collectively from the nephrons into the collecting duct system and ultimately is excreted as urine.Īcid-Base Balance: The acidity and alkalinity of blood is tightly regulated, and is slightly basic within a range of pH= 7.35-7.45. The nephron is the functional unit of the kidneys nephrons are contained within the renal medulla. Water moves into the compartment with a higher solute concentration or osmolality, until equilibrium is reached between the two compartments. 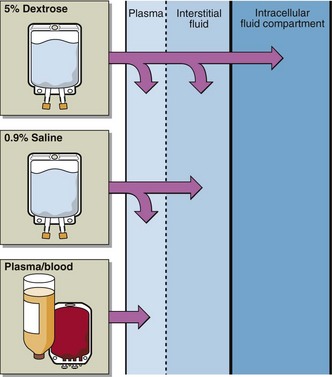
Intracellular fluid: Inside cells, cations (positively charged molecules): Ca2+, Mg2+, Na+ and anions (negatively charged molecules): Cl-, HCO-3 and proteins are in equal concentrations.Īs water and salt are ingested there is a change in the osmolality of the ECF, and subsequently water moves between the ECF and ICF, towards the higher solute concentration. Total body fluid: Total Body Water (TBW) = 60% x Body Weight.Įxtracellular fluid: is made up of Cations (positively charged molecules): Ca2+, Mg2+, Na+ and anions (negatively charged molecules): Cl-, HCO-3 and proteins are in equal concentrations. While both forces contribute to fluid movement across a capillary membrane, osmotic pressure is the major force that drives fluid across the plasma membrane of cells. 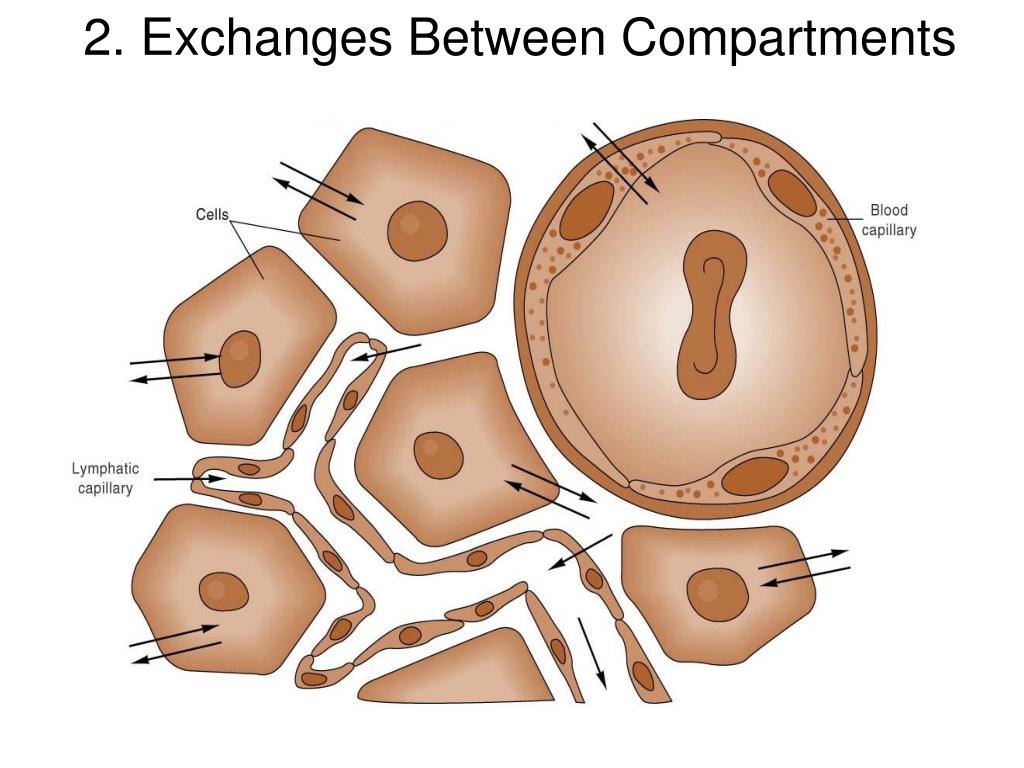
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
